Unlock new insights into muscle growth and tendon biology with myostatin cell lines. Discover how BPS Bioscience tools advance research in muscle-wasting diseases and regeneration.
Myostatin (MSTN), also known as GDF8 (growth differentiation factor 8), a member of the transforming growth factor-beta (TGF-β) superfamily, plays a crucial role in the regulation of skeletal muscle growth. It acts as a negative regulator by inhibiting myoblast proliferation and differentiation, making it a key player in muscle homeostasis. The discovery of myostatin has led to extensive research into its molecular mechanisms, with significant implications for muscle-wasting diseases, regenerative medicine, and athletic performance enhancement.
Recent advancements in cell line models, particularly the establishment of myostatin-knockout (Mstn-KO) C2C12 cell lines, have provided valuable insights into how myostatin controls muscle growth at the cellular and molecular levels. Moreover, emerging evidence suggests that myostatin is also involved in tendon biology, influencing tenogenic differentiation and musculoskeletal tissue interactions. This article explores the various aspects of myostatin's function, its impact on muscle and tendon differentiation, and the potential applications of a myostatin responsive cell line in research and medicine.
Myostatin as a Negative Regulator of Muscle Growth
Myostatin is primarily expressed in skeletal muscle and functions by inhibiting myoblast proliferation. Myostatin requires to activation, by proteolytic cleavage of the precursor protein. The first step in its activation is the removal of the signal peptide, followed by dimerization. The step includes furin, and the last step BMP-1/TLD. Studies using C2C12 myoblast cell lines have demonstrated that myostatin suppresses cell cycle progression by preventing the transition from the G1 to S phase. This is achieved through the upregulation of the cyclin-dependent kinase inhibitor p21, coupled with a reduction in the levels and activity of Cdk2 protein. This leads to the hypophosphorylation of the retinoblastoma (Rb) protein, resulting in cell cycle arrest at the G1 phase.
Myostatin can lead to muscle loss by either reducing protein synthesis or enhancing its catabolism. This occurs via inhibition of the AKT (protein kinase B)/mTOR (mammalian target of rapamycin) signaling pathway and promotion of transcription of atrogenes. It plays its roles by acting on SMAD and non-SMAD mediated pathways.
The consequences of myostatin inhibition have been observed in various animal models. Myostatin-null mice exhibit significantly increased skeletal muscle mass due to both hyperplasia (an increase in muscle cell number) and hypertrophy (an increase in muscle cell size). Similarly, cattle breeds such as the Belgian Blue and Piedmontese, known for their "double-muscling" phenotype, carry naturally occurring mutations in the myostatin gene, leading to dramatic muscle growth. These findings underscore myostatin's role as a key regulator of muscle mass and have sparked interest in therapeutic approaches aimed at blocking its activity to counteract muscle-wasting conditions.
Myostatin and Tenogenic Differentiation
Beyond its established role in muscle growth, recent studies suggest that myostatin also influences tendon biology. Myostatin has been found to promote the differentiation of C2C12 myoblasts into tenocytes (tendon-forming cells) via Smad3-mediated signaling.
During tenogenic differentiation, myostatin increases the expression of tenomodulin, a marker associated with tendon tissue, while simultaneously inhibiting myotube formation. This suggests that myostatin has a dual function: while it suppresses myogenesis (muscle formation), it enhances tendon differentiation. The activation of Smad3 signaling by myostatin appears to be a crucial mechanism driving this process.
These findings have important implications for tendon repair and regeneration. Since tendons have limited regenerative capacity due to their hypocellular and hypovascular nature, understanding myostatin’s role in tenocyte differentiation could lead to novel therapeutic strategies for tendon injuries. Future research may explore whether modulating myostatin expression can enhance tendon healing and improve musculoskeletal integration.
An understanding of Myostatin role by studies in myostatin-Knockout (Mstn-KO) Cell Lines
To further understand myostatin’s regulatory mechanisms, researchers have developed myostatin-knockout (Mstn-KO) C2C12 cell lines using CRISPR-Cas9 gene-editing technology. These genetically modified cells lack functional myostatin, providing a valuable tool for studying its role in muscle physiology.
Mstn-KO cell line were found to have:
- Enhanced Cell Proliferation:
- Mstn-KO cells exhibit significantly increased proliferation rates compared to wild-type C2C12 cells. This aligns with observations in myostatin-null animals, where the absence of myostatin leads to muscle overgrowth.
- Altered MicroRNA Expression:
- Real-time PCR (polymerase chain reaction) analysis has shown that several muscle growth-related microRNAs (miRNAs) are upregulated in Mstn-KO cells. Notably:
- miR-1, miR-206, and miR-133a (associated with myogenesis) are significantly increased.
- miR-23a (linked to muscle atrophy resistance) is also upregulated.
- miR-486 (involved in muscle homeostasis) is downregulated.
- These findings suggest that myostatin exerts its effects, at least in part, through miRNA-mediated pathways
- Real-time PCR (polymerase chain reaction) analysis has shown that several muscle growth-related microRNAs (miRNAs) are upregulated in Mstn-KO cells. Notably:
- Potential for Drug Discovery:
- The Mstn-KO C2C12 cell line provides a model for testing compounds that modulate muscle growth. Since myostatin inhibitors are being explored as treatments for muscle-wasting conditions such as muscular dystrophy and sarcopenia, these cell lines offer a platform for screening new therapeutic candidates.
Therapeutic Implications of Myostatin Research
Given its critical role in muscle regulation, myostatin has become a target for therapeutic intervention in various conditions, including:
- Muscular Dystrophy & Muscle-Wasting Disorders: Inhibition of myostatin is being investigated as a potential treatment to promote muscle regeneration in diseases such as Duchenne muscular dystrophy (DMD) and amyotrophic lateral sclerosis (ALS).
- Aging and Sarcopenia: Age-related muscle loss (sarcopenia) is a major health concern. Myostatin inhibitors could help counteract muscle decline and improve mobility in the elderly.
- Athletic Performance Enhancement: Although controversial, myostatin inhibitors have been explored for their potential to enhance muscle mass and strength in athletes. However, ethical and safety concerns remain.
- Tendon Repair and Regeneration: The discovery that myostatin influences tendon differentiation suggests that modulating its activity could improve tendon healing, particularly in conditions like rotator cuff injuries and Achilles tendon ruptures.
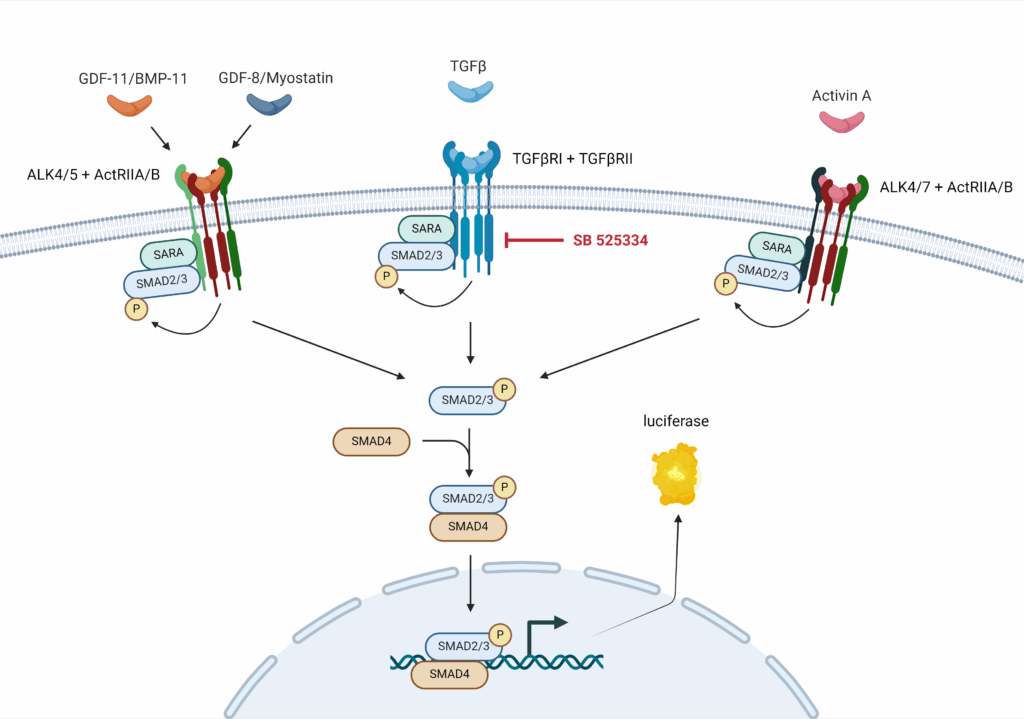
BPS Bioscience TGFβ/Activin A/Myostatin-Responsive Luciferase Reporter HEK293 Cell Line as a tool for myostatin related research
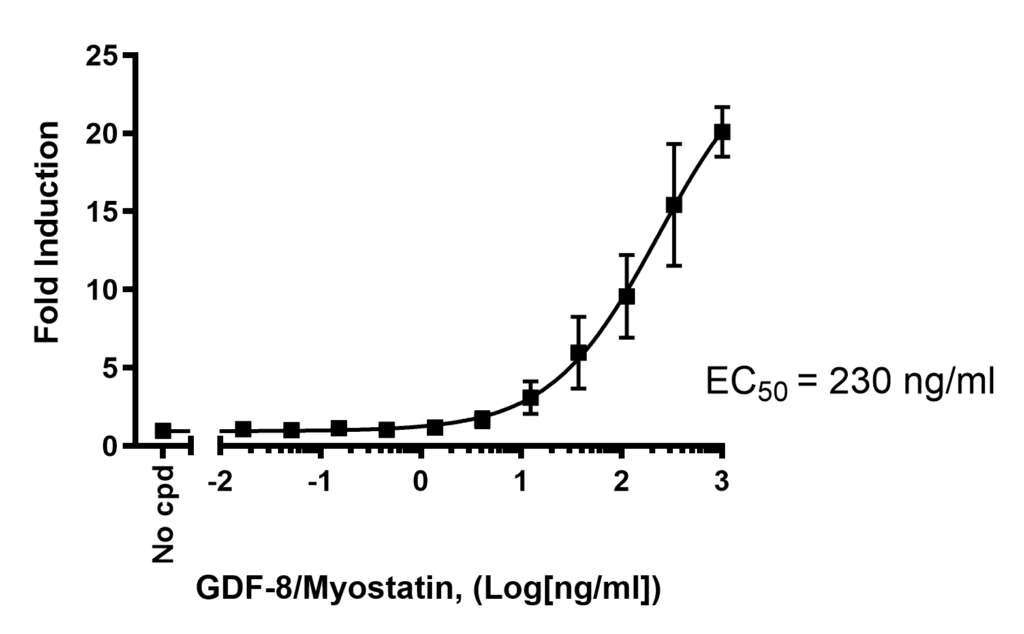
BPS Bioscience has developed a cell model where Firefly luciferase reporter is under the control of SMAD-responsive elements (SMAD binding elements, SBE). This cell line allows to monitor the activity of the TGFβ (transforming growth factor beta)/SMAD signaling pathway, and has been extensively validated. It is responsive to myostatin, allowing the study of inhibitors and activators of the myostatin related pathway.
Conclusion
Myostatin plays a central role in regulating skeletal muscle growth, with emerging evidence highlighting its involvement in tendon biology. Advances in genetic engineering have enabled the creation of myostatin-knockout cell lines, providing a powerful tool for investigating the molecular mechanisms of muscle and tendon development.
These insights open new avenues for therapeutic interventions in muscle-wasting diseases, age-related sarcopenia, and tendon injuries. As research continues, the potential to manipulate myostatin signaling for clinical benefit remains an exciting frontier in regenerative medicine.
Proteins & enzymes | Antibodies | Lentiviruses | Cell Lines | Biochemical and Cellular Assay Kits